Workdone for adiabatic3/15/2023 
Another example, when sound travels in a gas, there is no time for heat conduction in the medium, and so the propagation of sound is adiabatic.ģ. Another example, the adiabatic flame temperature uses this approximation to calculate the upper limit of flame temperature by assuming combustion loses no heat to its surroundings. The same can be said to be true for the expansion process of such a system. Even though the cylinders are not insulated and are quite conductive, that process is idealized to be adiabatic. Some chemical and physical processes occur too rapidly for energy to enter or leave the system as heat, allowing a convenient "adiabatic approximation" For example, the compression of a gas within a cylinder of an engine is assumed to occur so rapidly that on the time scale of the compression process, little of the system's energy can be transferred out as heat to the surroundings. Unlike an isothermal process, an adiabatic process transfers energy to the surroundings only as workĢ. Various dimensions of an adiabatic processġ.A process without transfer of heat or matter to or from a system, so that Q = 0, is called adiabatic, and such a system is said to be adiabatically isolated. Where α is the number of degrees of freedom divided by two, R is the universal gas constant and n is the number of moles in the system (a constant). For an ideal gas ( ideal gas law PV = nRT) the internal energy is given by It is desired to know how the values of dP and dV relate to each other as the adiabatic process proceeds. However, P does not remain constant during an adiabatic process but instead changes along with V. Pressure–volume work δW done by the system is defined as Any work (δW) done must be done at the expense of internal energy U, since no heat δQ is being supplied from the surroundings. Where dU is the change in the internal energy of the system and δW is work done by the system. Then, according to the first law of thermodynamics, The definition of an adiabatic process is that heat transfer to the system is zero, δQ = 0. In nature, this ideal kind occurs only approximately because it demands an infinitely slow process and no sources of dissipation. In one such kind, no entropy is produced within the system (no friction, viscous dissipation, etc.), and the work is only pressure-volume work (denoted by P dV). 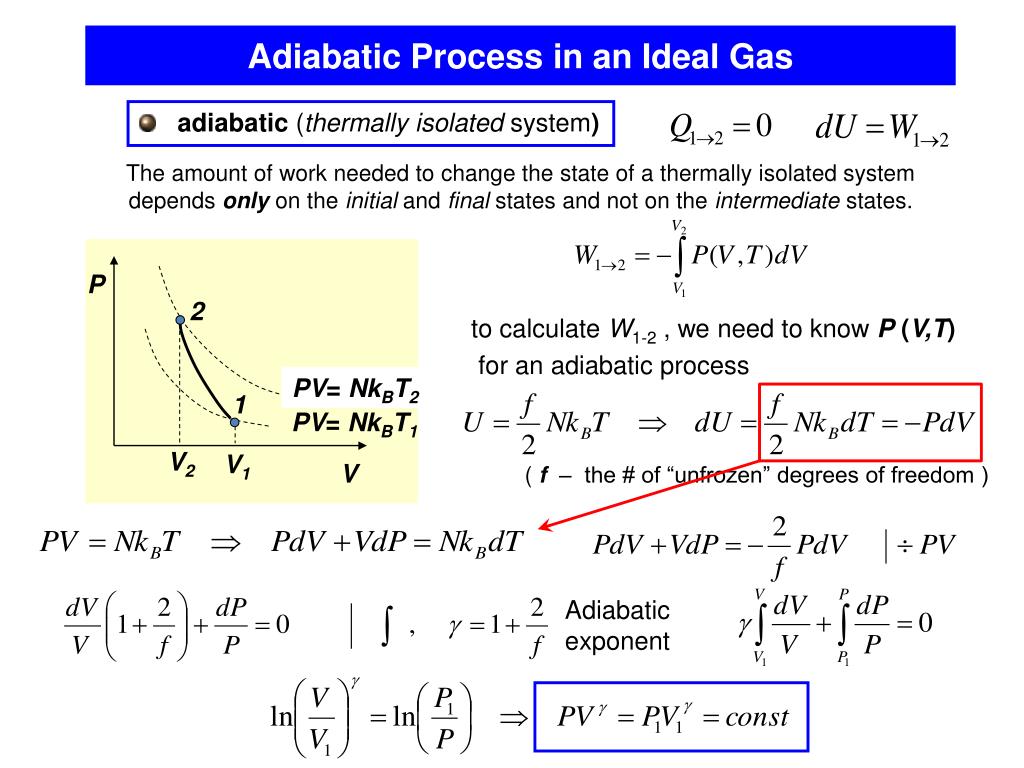
The transfer of energy as work into an adiabatically isolated system can be imagined as being of two idealized extreme kinds. For a closed system, one may write the first law of thermodynamics as ΔU = Q – W, where ΔU denotes the change of the system's internal energy, Q the quantity of energy added to it as heat, and W the work done by the system on its surroundings.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
